為什麼選擇XEOMIN®?
銷量No.1#
香港唯一^純淨+
零雜質*肉毒桿菌素
零雜質*肉毒桿菌素
有效減低出現
抗藥性風險1-2
抗藥性風險1-2
擁有多個國際認證
全球超過70+個國家批准用於醫學美容
超過98%改善度4
效果3-4天內見效5-6
效果3-4天內見效5-6
零雜質*的純淨
肉毒桿菌素有什麼好處?
零雜質*的純淨+肉毒桿菌素有什麼好處?
Z
ZERO 零雜質*減少產生抗體風險
E
EFFICACY 療效通過臨床認證
R
REDUCED 減低肉毒桿菌素產生抗藥性的風險1-2
O
OPTIMAL 每次療效均達致理想效果

你知道產生抗體的長遠影響嗎?

你知道產生抗體的長遠影響嗎?
1
美容用途
肉毒桿菌素屬於肌肉阻斷劑,當出現抗體導致療程失效,當然不能再達到減淡皺紋或其他美容目的。
2
醫療用途
肉毒桿菌素不只醫美,亦適用於醫療用途。因此一旦產生抗藥性,則可能影響將來醫學治療,構成長遠健康風險,情況可以相當嚴重。
德國製造的XEOMIN®是香港唯一^零雜質*純淨+的肉毒桿菌素品牌,能減少產生抗體的風險1-2,持續保持療程美容效果,同時降低療效遞減的機會1-2。所以在進行療程時,應選從一開始選擇純淨+零雜質的去皺針,美麗健康同時擁有!
XTRACT™ 先進雙重淨化技術
由德國百年藥廠Merz Aesthetics®利用先進的雙重淨化技術提煉而成,除去所有雜質*,只保留活性神經毒素,以減低身體產生抗體風險1-2,是在監管嚴格的GMP藥廠環境內生產之唯一不含雜質*的純淨+肉毒桿菌素產品。
A型肉毒桿菌素
雜質*
活性神經毒素
XEOMIN®
純活性神經毒素
純活性神經毒素
經淨化後的XEOMIN®能有效阻斷引致肌肉收縮的神經細胞訊息傳遞功能,使引致動態皺紋的肌肉得以放鬆,令面部線條變得平滑,並可防止新皺紋的形成。

XEOMIN® 適用位置
53.5%
超過98%的使用者
對XEOMIN®療程表示有改善。
對XEOMIN®療程表示有改善。
改善效果於療程後3 - 4天內5-6發揮作用
一般可維持4 - 6個月5-6
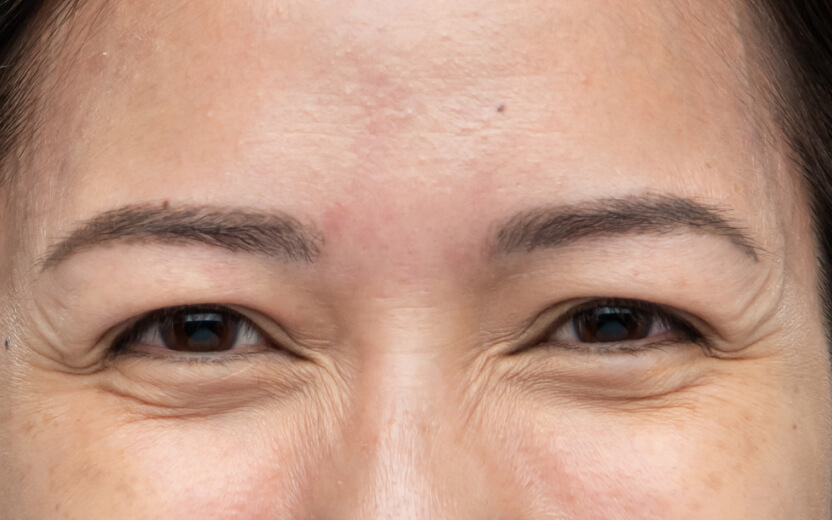
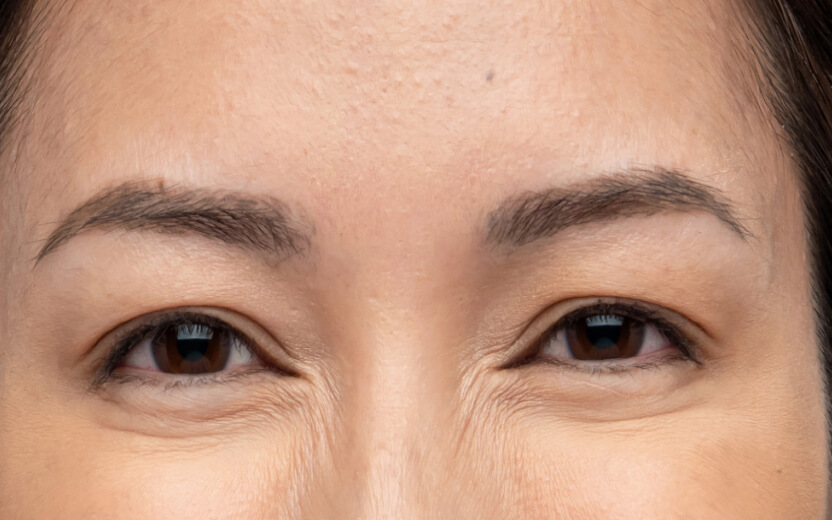
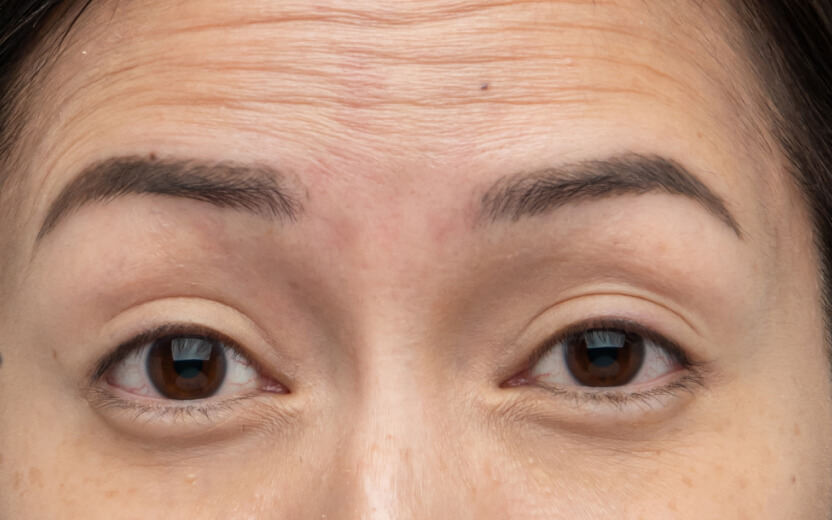
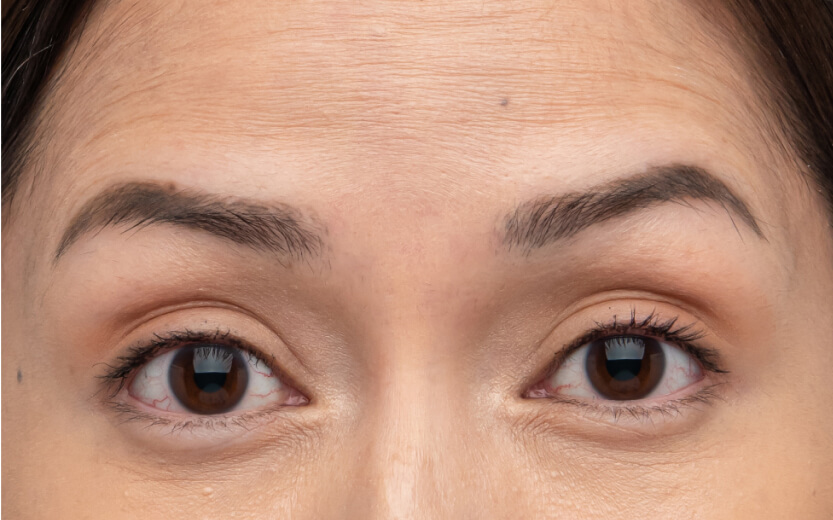
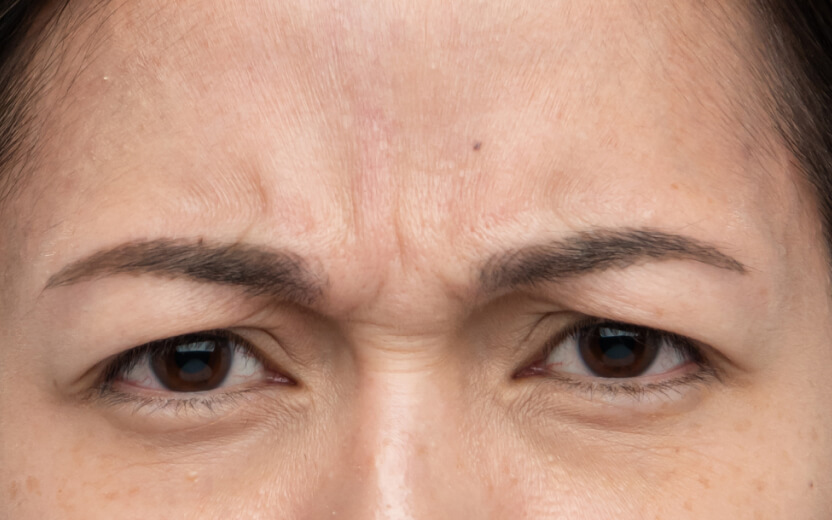
使用XEOMIN®的療程效果13
抬頭紋
眉心紋
魚尾紋
以上為真實個案,療程實際效果及維持時效因人而異,詳情請向醫生及醫護人員查詢。
你覺得肉毒桿菌素
療程效果有減退嗎?
療程效果有減退嗎?
可能是因為對肉毒桿菌素產生抗藥性!
你知道嗎?
%
香港用家
香港用家
曾感到療效遞減14
(2024年對比2021年增加4%)
%
香港用家
香港用家
希望在接受第一次肉毒桿菌素療程前被告知產生抗藥性+零雜質1-2的風險14, 以減低產生抗體和療程失效風險14。
%
香港用家
香港用家
如果可以重新選擇,用家願意重新選擇純淨+零雜質*的肉毒桿菌素,以減低產生抗體和療程失效風險14
現時市面部分傳統的肉毒桿菌素品牌含有雜質*,這些雜質*進入人體後會激活免疫系統、產生出抗體抗衡,久而久之便會出現抗藥性1-2。加上現時瘦臉、瘦小腿等熱門療程,普遍注射劑量偏高、亦加劇產生抗藥性的風險。情況如同攝取咖啡因,雖然少量攝取能幫助保持清醒、對抗疲勞,但當長期大量攝取,身體就會開始適應、令提神功效逐漸減弱。
一項數據調查指出14,於2021年及2024年訪問香港用家是否經歷療效遞減,結果發現在2021年已有7成用家感受到療效遞減、 到2024年更上��升至8成用家!而受訪者亦分別認為自己出現了不同程度的療效遞減跡象, 當中包括有療效持續時間短暫、療效不及之前、甚至完全感受不到療效!
有香港本地個案分享,指自己在接受了4次療程後,就發現療效急速減弱。經過多方查證後才明白自己正經歷肉毒桿菌素抗藥性,感到十分震驚!而且更擔心會影響往後的醫療用途、對自己健康構成風險;更覺得若能時光倒流,最希望能在療程前了解抗藥性問題,在決定進行療程前就會更加考慮清楚!
選用純淨+、零雜質*的肉毒桿菌素
* Impurities refer to the presence of complexing proteins, inactive neurotoxin, flagellin and bacterial DNA contaminants.1 / + Pure refers to the absence of complexing proteins, inactive neurotoxin, flagellin and bacterial DNA contaminants.1 It also contains immunologically inert excipients.2,3 / ^ 與其他香港註冊肉毒桿菌素品牌比較1,2 ; 香港註冊藥劑製品: www.drugoffice.gov.hk/ # No.1 Imported brand in Korea. Based on MFDS's 2020 Total Imports (50U + 100U) Record Data / 1. Park JY, et al. Neurotoxin impurities: A Review of Threats to Efficacy. Plast Reconstr Sur Glob Open.2020;8(1):e2627 / 2. Martin, M.U.; Frevert, J.; Tay,C.M. Complexing Protein-Free Botulinum Neurotoxin A Formulations: Implications of Excipients for Immunogenicity. Toxins 2024, 16, 101. / 3. Martin MU, Frevert J, Park J-Y, Cui H, Curry A, Loh WQ. Immunological Considerations of Polysorbate as an Excipient in Botulinum Neurotoxin Type A Formulations: A Narrative Review. Toxins. 2025;17(12):598 / 4. Imhof M, Kühne U. A Phase ilI Study of IncobotulinumtoxinA in the Treatment of Glabellar Frown Lines. 2011; 4(10):28-34 / 5. Prager W, et al. Clin Interv Aging. 2013;8:449-56 6. Rappi T, et al. Clin Cosmet Investig Dermatol. 2013; Sep 24: 6:211-19 / 7. Kane MA, et al. Dermatol Surg. 2015;41(11):1319. / 8. Sattler G, et al. Dermatol Surg 2010;36(Suppl 4):2146–54 / 9. Prager W, et al. Dermatol Surg 2010; 36(Suppl 4):2155–60. / 10. Fischer T, et al. J Drugs Dermatol. 2020;19(5):461-469. / 11. Chao YY, et al. J Clin Aesthet Dermatol. 2019;12(12):E53-E57 / 12. Saybel A, et al. J Drugs Dermatol 2015;14(11):1291- 1296. / 13. XEOMIN® Approved Product Information. Aug 2021. / 14. 資料來源:根據2024年一項由Merz Aesthetics聯同Frost & Sullivan 於9個亞太地區進行關於「肉毒桿菌素抗藥性」的消費者研究調查,合共2,588名使用肉毒桿菌素的受訪對象 (曾注射三次或以上含雜質的肉毒桿菌素),年齡介乎21至55歲。
尋找獲認證的醫療中心